AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Osmos 5 download free12/29/2023 
across the membrane and that therefore the P.D.  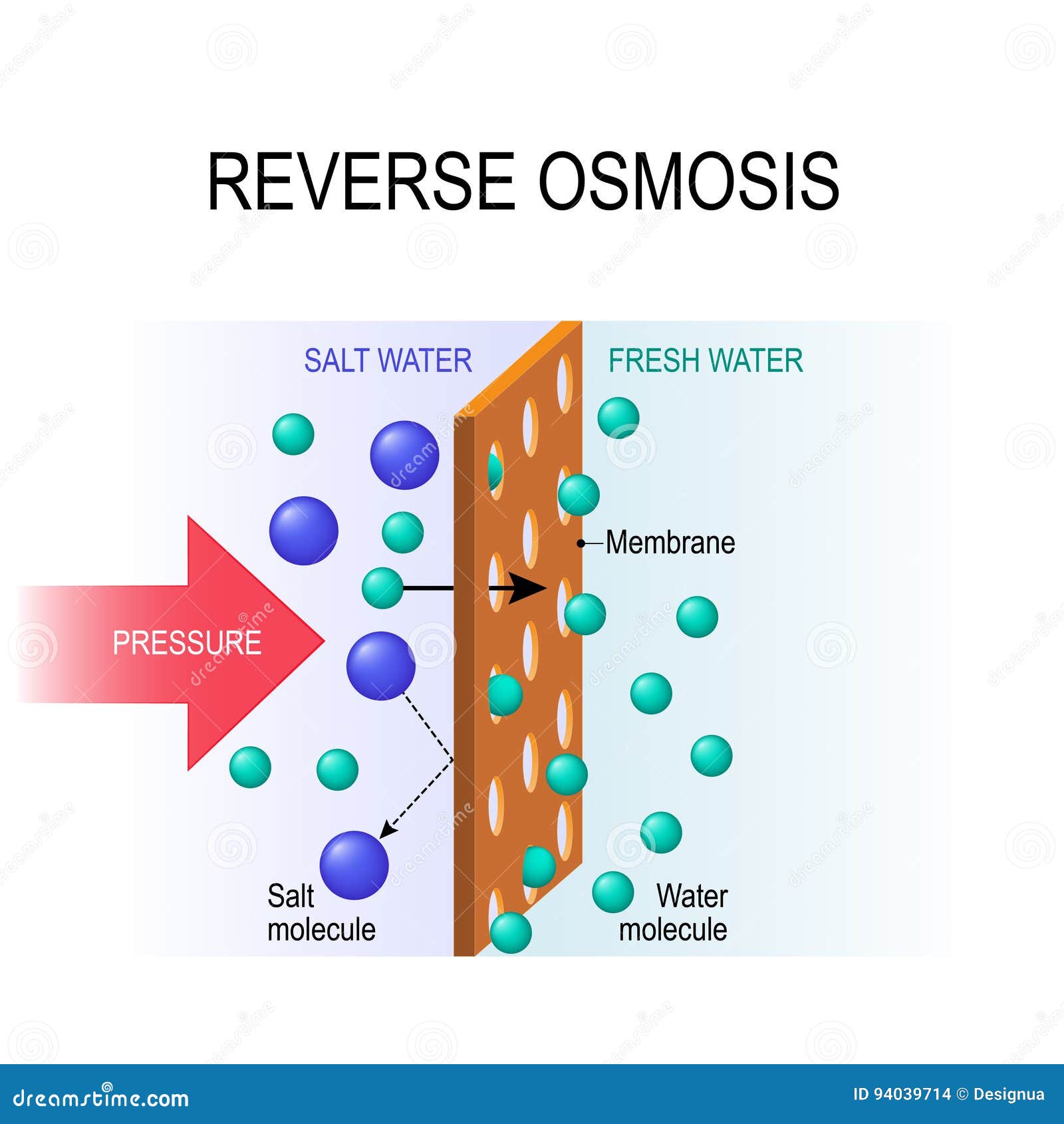
It is shown, however, that the diffusion potential without interposition of the membrane differs in a definite sense from the P.D. It is shown that the hydrogen ion concentration of the solution affects the transport curves and the diffusion potentials in a similar way. This makes it probable that the driving force for the electrical transport of water from the side of pure water into solution is primarily a diffusion potential. It is also shown that a similar valency effect exists in the diffusion potentials between salt solutions and pure water without the interposition of a membrane. across the membrane ( E) are given, showing that when an electrical effect is added to the purely osmotic effect of the salt solution in the transport of water from the side of pure water to the solution, the latter possesses a considerable negative charge which increases with increasing valency of the anion of the salt and diminishes with increasing valency of the cation. It had been shown in previous papers that when a salt solution is separated from pure water by a collodion membrane, water diffuses through the membrane as if it were positively charged and as if it were attracted by the anion of the salt in solution and repelled by the cation with a force increasing with the valency.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
